Call: 08045804767
IVF Disposable
IVF Disposable Specification
- Shelf Life
- 2 Years
- Usage Type
- Single Use
- Storage Instructions
- Store in Cool Dry Place
- Instruments Type
- Laboratory Disposables
- Accuracy
- High (Process Dependent)
- Function
- Sample Collection and Handling
- Equipment Type
- IVF Disposable
- Material
- Medical Grade Plastic
- Condition
- New
- Technology
- Sterile Manufacturing
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- Laboratory Procedures in IVF
- Power Source
- None
- Dimension (L*W*H)
- Standard Sizes (Varies by Type)
- Weight
- Lightweight
- Color
- Transparent
- Shape
- Round and Flat (Type Dependent)
- Packaging
- Individually Packed
- Toxicity
- Non Toxic
- RNase/DNase Free
- Yes
- Sterility
- Gamma Sterilized
- Bio-compatibility
- Certified Biocompatible
- Intended Application
- Human IVF Lab Procedures
About IVF Disposable
Falcon IVF DisposableCertified sterile, non pyrogenic and non embryonic. Each lot is tested for embryo toxicity using a two celled mouse embryo test. These plastic wares are tested by culturing embryos both in plastic ware and media that have been in contact with the lab ware for 24 hours at 370C.
Advanced Sterility and Safety
Each IVF Disposable undergoes gamma sterilization and is individually packed to assure maximum sterility. The products are manufactured under strict sterile conditions, ensuring they are RNase/DNase free, non-toxic, and fully biocompatible. These qualities support safe and reliable performance in sensitive IVF laboratory procedures.
Versatile Design for Optimal Performance
Our disposables come in both round and flat shapes, tailored to different procedural requirements in the IVF process. Made from medical grade plastic and designed for single-use, these items minimize contamination risk, while their high accuracy and standardized sizes enhance laboratory precision and workflow.
FAQs of IVF Disposable:
Q: How do IVF Disposables ensure sterility and safety during IVF lab procedures?
A: IVF Disposables are gamma sterilized and individually packed in a sterile environment, guaranteeing that each item is RNase/DNase free and non-toxic. Their biocompatibility certification assures safe handling of human samples during IVF procedures.Q: What are the recommended storage conditions for these disposables?
A: The disposables should be stored in a cool and dry place to maintain their sterility and extend their shelf life, which is up to two years from the date of manufacture.Q: When should these IVF disposables be used?
A: These products are intended for single use during laboratory procedures in human IVF labs, particularly for the collection and handling of samples, where sterility and accuracy are essential.Q: Where can these disposables be utilized most effectively?
A: They are best used in clinical and laboratory environments specializing in human in-vitro fertilization procedures, ensuring sample integrity and operator safety.Q: What process is followed to guarantee the quality and sterility of the IVF Disposables?
A: The manufacturing process uses advanced sterile technology and medical grade plastic, with all disposables undergoing gamma sterilization. Each item is individually inspected and packed to meet high quality standards.Q: How does the design of these disposables benefit laboratory personnel?
A: The lightweight, portable, and transparent design makes handling and observation easier, while the variety of shapes allows for optimal suitability to different laboratory tasks, improving workflow and accuracy.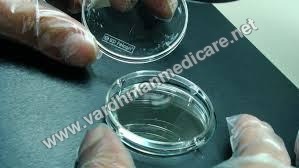
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Infertility Care Category
Klenzaids Test Tube Warmer
Voltage : 220240 V AC, 50 Hz
Wall Mounted : No
Power Consumption : 60 Watts
Technology : Electric Heating
Ovum Aspiration Pump
Price 270000.0 INR / Piece
Minimum Order Quantity : 1 Piece
Voltage : 220 Volt (v)
Wall Mounted : No
Power Consumption : 200 Watt (W)
Technology : Vacuum
Heating Stage (Tokai Hit, Japan)
Voltage : 110/220V
Wall Mounted : No
Power Consumption : 40W
Technology : Precision Temperature Control, Digital HUD
IVF Lab Pressurising Module
Price 150000 INR / Piece
Minimum Order Quantity : 1 Piece
Voltage : 230 volts, 50 Hzs Volt (v)
Wall Mounted : No
Power Consumption : 750 Watt (W)
Technology : Latest technology

 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry




